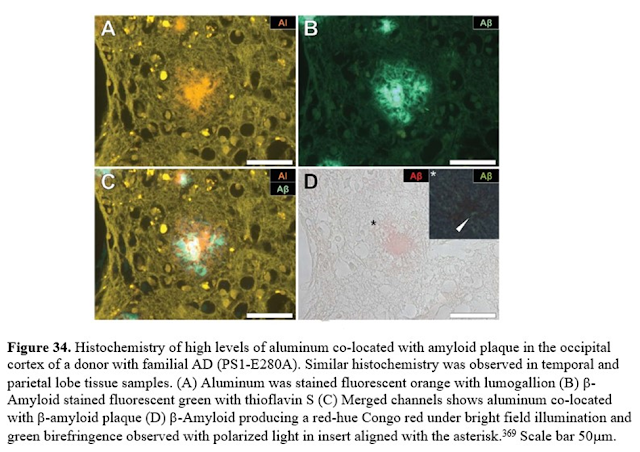
Finding
a Cause and Potential Cures for Alzheimer’s Disease
Climbing
the Ladder of AD Causation
Publication Spring 2022 available on Amazon
Author: Dr. Dennis N. Crouse, BSc Biochemistry, -
Harvard College, Ph.D. Organic Chemistry - Harvard University Chemistry
Department, Post-graduate courses:
Understanding Dementia - Wicking Faculty of Health, University of
Tasmania, Fundamentals of Neuroscience - Harvard.
Introduction
Alzheimer’s disease (AD) is prevalent in the U.S. with
an estimated 6.2 million people age 65 and older currently living with AD. Unfortunately,
my mother is one of those people. Her short-term memory was going from bad to
worse when she was 85. Her doctor was following this trend with the mini-mental
state exam (MMSE) and reported that she had MCI that could lead to AD. Several
years later, magnetic resonance imaging (MRI) of her brain indicated she had “accelerated
brain atrophy” that is a characteristic biomarker of AD used to diagnose AD.
Being trained in biochemistry and chemistry at Harvard
College and Harvard University, respectively, I decided to take action with the
goal of identifying causal factors of AD so a cure for mom might be found.
After several years of researching the scientific literature on AD, a causal
factor of AD was identified and a potential cure for mom was tried with some success!
Mom’s MMSE score improved and thankfully by age 89 she
could describe the daily news she read or heard. This potential cure for
some symptomologies of AD is also a preventative as documented in my 2016 book
titled: “Prevent Alzheimer’s, Autism, and Stroke with 7 Supplements, 7
Lifestyle Choices, and a Dissolved Mineral”211.
Most AD cases are sporadic and result from hereditary
and environmental causes. A subset (i.e., 66%) of sporadic AD cases, that are
usually diagnosed before age 70, are associated with a specific genotype (i.e.,
ApoE
e4
allele) increasing the risk of
AD and amount of cerebral beta-amyloid protein (Ab-42)178.
Less than 2% of total AD cases are familial early-onset AD (EOAD) that is associated
with mutations in presenilin 1 and 2 genes. EOAD is usually diagnosed before
age 65 and is also characterized as having an increased amount of Ab-42179. At least one third of AD
patients do not have an ApoE e4
allele. In addition, half
of those with two copies of the ApoE e4
allele, do not get AD and survive to age 80178. Also, 24% of people
with high
levels of Ab-42
do
not have in vivo biomarkers of AD180,181. Therefore, logically
there must be one or more environmental causes of sporadic AD that are made
worse by increased amounts of cerebral beta-amyloid Ab-42.
My mother has the ApoE e3/e4 alleles and I have the ApoE e2/e4 alleles. I
got the ApoE e4 allele from my mother and the ApoE e2 allele from my father who had ApoE e2/e3 alleles . Carriers
of the ApoE e4
allele have increased
odds of getting AD as compared to carriers of two ApoE e3 alleles, like my sister who has ApoE
e3/e3 alleles.
The odds of
getting sporadic AD are based upon environmental factors, age and sex of the
carrier, and if the carrier has one or two copies of the ApoE e4 allele (see two graphs below)178. At age 65 I had
approximately 2-fold greater odds of getting AD due to my genetics as compared
with my sister178. My mother’s odds of getting AD at age 65 were 4.5-fold
greater than my sister178. Men and women with ApoE e4/e4 alleles at age 60 have 11-fold and 12-fold
greater odds of getting AD, respectively178. But in spite of these greater
odds, studies of twin pairs have demonstrated the ApoE e4 allele accounts for only 10.7% of the variance
in Ab-42 accumulation, suggesting significant environmental
factor(s) as cause(s) of sporadic AD315-318.

Relative odds of getting AD
based upon Caucasian subjects in clinical and autopsy studies178
When I began researching AD in 2012 it became apparent
that the field had become dominated by some very large and financially powerful
players (e.g., aluminum industry, pharmaceutical industry and their partner the
Alzheimer’s Organization) They had decided for their own financial gain that scientists
in the U.S. and U.K. should play by their rule: if you are not working on decreasing
beta-amyloid protein (Ab-42)
you are not working on AD. In spite of their rule there are three proposed
theories, not just one, on the cause of AD:
·
Aluminum
Accumulation
·
Beta-amyloid
Accumulation
·
Calcium Dyshomeostasis
Playing by their rule required that you ignore two of
these theories and don’t work on hypothesizing a fourth or fifth theory. Being
close-minded does not facilitate finding cure(s) for a disease. Ironically at
an Alzheimer’s Organization talk in 2015 by Claudia Kawas on the 90+ study it
was pointed out those with just neuronal beta-amyloid accumulation do not have
a high risk of AD dementia. Two other cerebral pathologies are required to
significantly increase odds of AD dementia. A significant number of people are
resilient to beta-amyloid accumulation181.
Even before Claudia’s talk, I decided to not play by
their rule. I read research papers on all three proposed theories with the goal
of building a unified theory of AD.
Molecular epidemiological data is available that can
be used to find causal factors of AD and cures for AD. Looking for correlations in this data, revealed
a causal factor of AD. I found that there is molecular epidemiological data
providing convincing evidence that aluminum is both a causal factor of AD and
drinking silica rich water is a preventative intervention for AD and a
potential cure for some symptomology of AD.
Finding the
cause of a disease facilitates finding the cure and finding factors that mediate
or modulate the disease can reveal the cause of a disease. A literature search for
modulators of AD uncovered two independent French epidemiology studies
published in 2005 and 2008 that used two different data sets and surprisingly
found drinking water containing greater than 4mg/day of silica as orthosilicic
acid (OSA) or drinking water greater than 12mg/liter of OSA significantly
lowered the odds of getting AD200,209. With further searching I found
a small 2006 study where 3 out of 15 patients with AD had an improvement in
cognition after just 12 weeks of daily drinking OSA rich water42,43.
The 2008 French epidemiology study also found that aluminum levels in drinking
water of 100mcg/liter or more significantly increased the odds of getting AD200.
These studies were the primary inspiration for my first book211. My second
book looked at the health of people who drank OSA rich water for their entire
life239.
Does
drinking OSA rich water significantly lower the odds of getting AD in those,
like my mother and I, who are carriers of the ApoE e4 allele? This question was answered during the writing of my
second book when I discovered Ibadan, Nigeria. The drinking water of Ibadan has
a high level of OSA (i.e., 35ppm) compared with average level in the U.S. (i.e.,
11ppm)319,320.
From 1992 to
2006 a cohort of 2,245 elderly Nigerians living in Ibadan were genotyped and
clinically diagnosed. Also, a cohort of 2,147 elderly African
Americans living in Indianapolis, Indiana, were genotyped and diagnosed. In
this latter cohort, people with the ApoE e4 allele had increased odds of getting AD. In general people
living in Ibadan have 2-fold less risk of AD that those living in Indianapolis321,322.
Importantly, unlike the cohort of African Americans living in
Indianapolis, the cohort of people with the ApoE e4 allele
living in Ibadan did not have increased
odds of getting AD321,322. Therefore, drinking OSA rich water is
an environmental factor that significantly lowers the odds of getting AD even
in those with the ApoE e4 allele.
Based upon these
studies I began in September of 2015 drinking 4 cups a day of OSA rich water
(i.e., Silicade) spaced throughout the day. After 6 years I had my body burden
of accumulated aluminum tested and it was found to be in the range of a healthy
22-year-old. These test results made me feel much younger than my 75 years and also
made me confident that even with the ApoE e4
allele I would not get AD.
In addition
to aluminum there are many environmental factors that negatively impact
cognitive health. These environmental factors could also be potential causal
factors of AD. The scientific literature was searched without success for links
between these “brain drainers” and AD. This search revealed that essential
nutrients could be used to detoxify these brain drainers as summarized in my
third book titled “Increased IQ, Cognition, and Covid 19 Cure Rate with
Essential Nutrients”40.
In 2018 I
began reading about the new science of cause and effect called “causal inference”
and applying it to finding causes, mediators, and modulators of AD. Having
acquired a large amount of data on causal factors of AD, I found it could be
logically organized as a “ladder of AD causation” inspired by causal inference.
The result is a data-based logical argument for aluminum being the cause of AD based
upon the current scientific literature and is the subject of this my fourth
book.
Buy Book